Orion™: Bring comprehensive eye health into focus with innovative analysis software
▸ Analyze images and data from the entire spectrum of devices and platforms
▸ Attain best-in-class retinal segmentation with proven, AI-enhanced analysis
▸ Reduce read times with automated measurements, using intuitive software
“With [Voxeleron], we can start to say [to our sponsors] what do you want to know rather than what can you learn. This is really changing that paradigm. If you have a therapeutic that you would like to understand what part of the retina is being affected by it, we can now offer data to answer those questions.”
“We are thrilled to partner with Voxeleron for our device-agnostic ophthalmic imaging analysis needs. Orion’s comprehensive capabilities and seamless compatibility with diverse imaging modalities have transformed our clinical trial operations. The intuitive interface and prompt support from the Voxeleron team have streamlined our processes, reducing reading times and boosting overall efficiency.”
Why Choose Orion
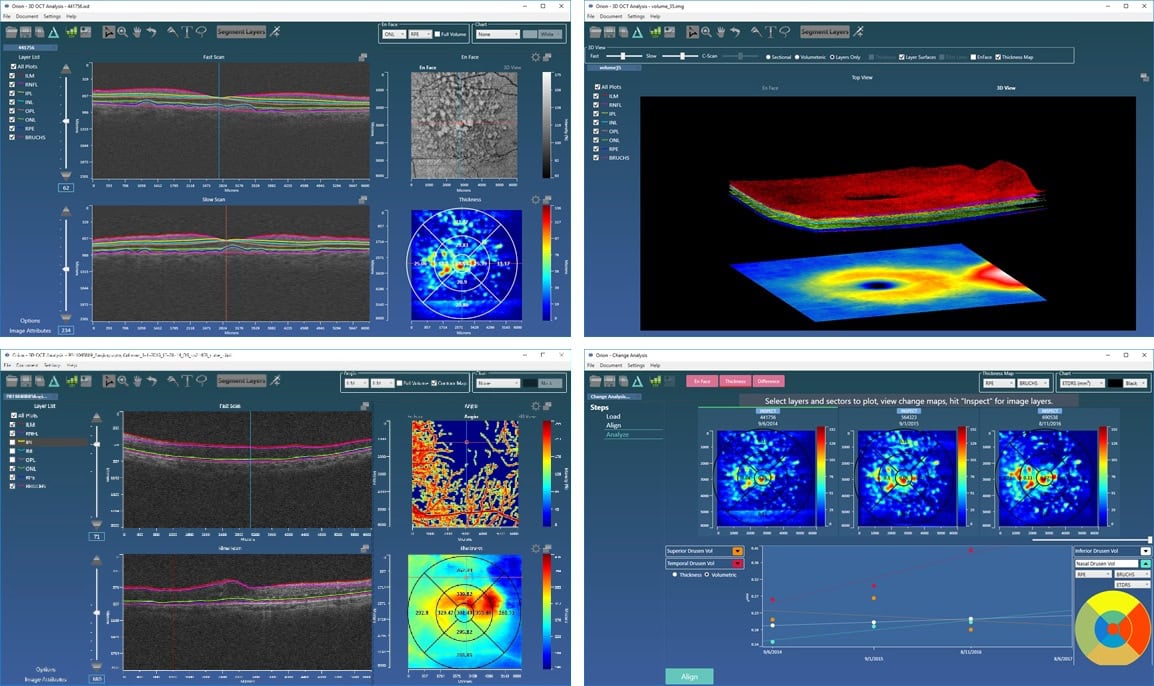
Device-independent 8 retinal layer segmentation software
The limited functionality of existing OCT tools has constrained analysis to only two or three retinal layers, despite the clinical importance of the inner retinal layers in disease detection and prognosis. We address this technology gap, providing researchers 8-layer segmentation and the needed biomarkers for early detection and prognosis of ocular and neurological diseases.
Orion advanced optical coherence tomography (OCT) analysis software is fully automated and entirely platform independent – truly vendor neutral – and processes data from all major OCT scanners in any of their formats. Orion provides 3D visualization, layer editing wizards, angiography support, batch processing and many more features.
Reading centers choose Orion to improve speed and accuracy, generate actionable results and, ultimately, drive better patient outcomes. *
*Orion has not been approved for clinical use. It is a research tool, and may not be used to support the treatment or diagnosis of human patients.
See how Orion seamlessly interfaces with iNebula™
Orion integrates with iNebula, our complete, cloud-based clinical trial platform. Image data and results are shared such that Orion opens data sent from iNebula and sends analysis results back, auto-populating an electronic case report form (eCRF) where provisioned. This seamless workflow is 21 CFR Part 11 compliant, fast, accurate and extensive in terms of the endpoints offered. In addition to that, the image data can come in any format and from any device!